Identification and Quantification of Active Pharmaceutical Ingredients (APIs)
Active Pharmaceutical Ingredients (APIs) are the active chemical components of a drug product that are responsible for the desired therapeutic effects of that drug.
Once an API is developed, it is combined with other inactive ingredients to create a final dosage form such as a tablet, capsule, or liquid that can be administered to patients.
The quality and purity of the API are critical to the safety and efficacy of the drug product; thus, the identification and quantification of active pharmaceutical ingredients (APIs) is a crucial step in the quality control of pharmaceutical products.
There are various methods available for this purpose, such as High-performance liquid chromatography (HPLC), Gas chromatography (GC), Mass spectrometry (MS), Fourier transform infrared spectroscopy (FTIR), Ultraviolet-visible spectroscopy (UV-Vis), Nuclear magnetic resonance (NMR) spectroscopy, X-ray diffraction (XRD), and High-performance thin layer chromatography (HPTLC).
Depending on the specific characteristics of the API and the dosage form, these techniques are used.
High-performance thin-layer chromatography (HPTLC) is a technique used for the identification and quantification of active pharmaceutical ingredients (APIs).
This technique is based on the separation of compounds based on their affinity for a stationary phase and a mobile phase.
The stationary phase in HPTLC is a thin layer of silica gel or cellulose coated on a glass plate, while the mobile phase is a solvent mixture that moves through the stationary phase via capillary action.
Learn about the Identification and Quantification of API’s
Identification of APIs by HPTLC
The identification of APIs by HPTLC involves the separation of the compound from the sample matrix and comparison with a standard compound.
The Rf value, which is the ratio of the distance traveled by the compound to the distance traveled by the mobile phase, is used to identify the compound.
The Rf value of the compound in the sample is compared to the Rf value of the standard compound, and if they match, then the compound is identified.
Quantification of APIs by HPTLC
The quantification of APIs by HPTLC involves the determination of the amount of the compound in the sample. This is done by comparing the peak area of the compound in the sample to the peak area of the standard compound.
The peak area is proportional to the amount of the compound, and therefore, the amount of the compound in the sample can be calculated.
HPTLC has high resolution, sensitivity, and reproducibility, making it an ideal method for the analysis of APIs. Also, the technique is cost-effective, making it advantageous over other techniques.
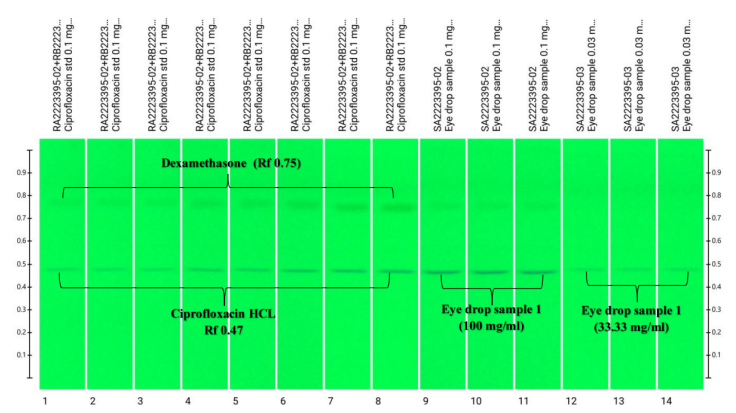
For example, identification and quantification of APIs of Ciprofloxacin and Dexamethasone in marketed opthamic solutions were carried out at our Anchrom Lab, Mumbai, by using the CAMAG HPTLC technique.
In the above developed Method, we have simultaneously quantified two APIs from the same marketed optamatic solution on a single plate. This demonstrates the effectiveness and versatility of HPTLC in the analysis of APIs.
HPTLC is an ideal method for analyzing APIs. It can also be applied in herbal drug testing to differentiate between related species.
Anchrom Enterprises Pvt. Ltd is a leading provider of HPTLC analysis services and HPTLC analytical instruments for plant extracts, drugs, cosmetics ingredients, and forensic science. Contact us at lab@anchrom.in for more information.